Pituitary Tumors
Background
Pituitary tumors like meningiomas account for 10% - 15% of all primary brain tumors. 75% of pituitary tumors are functional. They have a predeliction for females and are symptomatic. 70% occur between age 30 to 50.
MEN-1 and MEN 2 syndromes predispose to pituitary tumors. MEN 1 is associated with Pituitary, parathyroid and pancreas tumors and is associated with an autosomal dominant mutation 11q13 / menin.
The anatomy of the pituitary consists of the anterior axis which produces prolactin, growth hormone, ACTH, TSH, Lh AND FSH. The posterior produces ADH and oxytocin. The anterior pituitary arises from Rathke's pouch, the posterior is an extension of the third ventricle and it is located in the sella turcica.
The cavernous sinus is nearby. CN III-IV, V.1 - V.2 and VI are found in the cavernous sinus
Cells of non-functional tumors are chromophobic cells. For functional tumors the basophilic cells produce ACTH, TSH, LH and FSH. The acidophilic cells produce GH and PRL.
The most common functional pituitary tumors in order of appearance are:
- Prolactinoma at 30% and then
- Growth hormone at 25% and then
- ACTH at about 15%.
The most common pituitary tumors in males and the elderly are nonfunctioning or growth hormone producing tumors. In females, PL and ACTH are more common. Macroadenomas are more common than microadenomas overall, but microadenomas appear more often in females than males.
Microadenomas are the most common pituitary tumor in females, particularly PRL secreting tumors. Calcium deposits and amyloid are prominant on microscopic examination of prolactinomas. Pituitary adenomas frequently stain for synaptophysin, chromogranin, and hormone specific stains.
Workup and Staging
Pituitary tumors are found in close proximity to the optic chiasm and cavernous sinus. Physical symptoms relate to mass effect in these regions. Secreting pituitary adenomas produce symptoms related to their activity (growth hormone, galactorrhea, amenorrhea). The most common physical findings are bitemporal hemianopsia, headache and occulomotor deficits as well as V.1/V.2 deficits.
The workup includes H&P including CN assessment, visual fields, endocrine disorder signs. Lab studies including hormone levels: GH, PL, ACTH, TSH. An MRI with thin slices should be obtained and any mass biopsied via the trans-sphenoidal aproach.
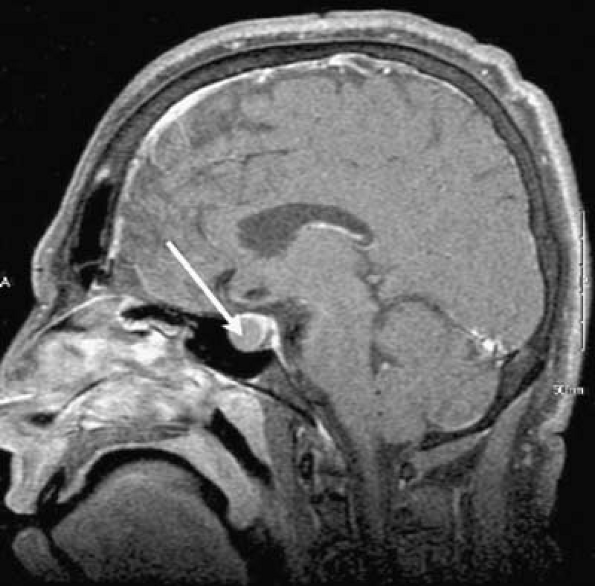 |
Pituitary masses can be craniopharyngiomas, meningiomas, gliomas, suprasellar germ cell tumors, or metastases. They can also be benign lesions.
Prolactinomas present with galactorrhea, amenorrhea, decreased libido adn infertility. PL levels are elevated > 20 - 25. Medical therapy for prolactinomas includes the use of bromocriptine.
TSH secreting tumors have a high rate of recurrence after resection. Increased risk factors include a history of thyroid ablation, Hashimoto's disease, prior radiation or surgery.
Growth hormone tumors present with acromegaly, gigantism in children.
ACTH presents as Cushing's disease. Cushings Disease includes round facies, central weight gain, hyperhidrosis, telengiectasia, thinning of the skin. It is related to high cortisol levels caused by increased ACTH production. Laboratory testing includes the dexamethone suppression test. This test should reduce cortisol levels when low dose dexamethasone is administered.
Most non-secretory tumors present with mass effect symtoms: visual field cuts, occulomotor defects and disconjugate gaze or diploplia, or hypopituitism.
Treatment and Prognosis
There are two general treatment paradigms:
- Surgical resection (transsphenoidal) if hypersecreting or symptomatic for non-secreting tumors → observation or post-op radiation therapy
- Definitive Radiation therapy to the tumor + 1.5 cm margin to 45 - 50.4 Gy (no gross disease) to 54 Gy if gross disease.
This same algorithm is used in non-functioning (ie non-secreting pituitary adenomas. The hormone normalization rate in secreting tumors normalizes 70% - 80% after surgery but the condition returns and the ultimate normalization rate is 30% - 40% with time.
For prolactinomas the general treatment begins with initial treatment with bromocriptine. Pharmacologic suppression of prolactinomas usually takes 1 - 2 months. Many have intolerance to bromocriptine due to nausea, headache and fatigue.
Surgery
Transsphenoidal microsurgery for microadenomas is the preferred treatment. This allows decompression, and debulking of larger tumors and reduction of hyperfunctioning tumors. For larger tumors, a frontal craniotomy approach is used, especially when there is cavernous sinus invasion or frontal lobe involvement. There is no visible scar with the trans-sphenoidal approach.
Pituitary adenomas are divided by size:
- microadenoma < 1 cm
- macroadenoma ≥ 1 cm
- picoadenoma < 0.3 cm
Surgery gives a 95% local control after transsphenoidal resection with better control for microadenomas after resection.
Tumors > 2 cm, high preoperative prolactin levels advanced age and longer duration of amenorrhea are poor prognostic signs after transsphenoidal resection. For tumors secreting growth hormone, advers prognostic signs include high pre-op Growth hormone levels and somatomedin C levels, as well as tumors > 1 cm and extrasellar extension.
Radiation Therapy
Radiation therapy may be used as definitive treatment or for post-operative adjuvant treatment.
The primary indications for radiation therapy include:
- Medically inoperable tumors
- Persistence of hormone defect after surgery
- Macroadenoma with STR or decompression only surgery
- Post-operative recurrence
Dose data supports at least 45 Gy. This is old data from the University of Florida from 1991. The 10 year local control rate was 95%. Stereotactic Radiosurgery is commonly used in pituitary microadenomas. SRS yields better hormonal secretion control than
Radiation therapy provides best outcomes for GH secreting tumors, followed by ACTH secreting tumors, finally prolactinomas.
- GH control > 80%
- ACTH control 50% - 80%
- PL control 30% - 40%
Medical treatment (bromocriptine, octreotide, somatostatin) should be discontinued before radiation therapy. Radiation sensitivity is lowered with adjuvant medical treatment.
Hormone normalization after radiation therapy may take years. Growth hormone secreting tumors normalize at 2 - 5 years for 50%, 70% after 10 years.
Stereotactic radiosurgery doses vary based on whether or not the tumor is functional or not:
- Functional Tumor SRS dose: 20 Gy
- Non-functional tumor SRS dose: 14 - 16 Gy
Gamma knife has less homogenous dosing than clinac based SRS, but more precise setup and slightly less normal tissue treated. Similar conformality and outcomes are acheived with either technique at a substantially less cost with the clininac based treatments.
Fractionated SR is preferred when the pituitary lesion is > 3 cm and/or the lesion is < 2 mm from the chiasm.
Medical Management
Pharmacologic agents used in the treatment of GH secreting tumors include somatostatin, octreotide, pegvisomant.
ACTH tumors are treated with ketoconazole (best treatment). Other treatments include mitotane, RU-486, metyrapone and cyproheptadine.
Toxicity
TD 5/5 dose for hypopituitarism is 40 - 45 Gy. Growth hormone levels decrease first followed by LH/FSHJ → TSH/ACTH.
Optic nerves and chiasm dose limits are 50 - 54 Gy for fractionated radiation and 8 Gy for single fraction radiosurgery. The main benefits of SRS include decreased neurocognitive sequalae, and possible preservation of normal pituitary function, due to reduced dose to hypothalamus. This comes at an increased risk of damage to the chiasm and optic nerves.
The best measure of RT dose response in GH secreting tumors is to monitor IGF-1 (insulin like growth factor -1 ) levels. GH is the first hormone to decrease following radiation therapy.
Operative mortality rates are low at 1% - 2%. The operative complication rate is 15% - 20%. Diabetes insipidus is the most frequent post surgical complication followed by hyponatremia then CSF leak.
Men with GH secreting pituitary adenomas are prone to increased rates of second malignancies after radiation therapy.